New Insights into Obesity Management: Chchd10 Protein's Crucial Role in Adipose Tissue Regulation Revealed
2025-03-17
Author: Wei Ling
Introduction
Obesity continues to pose a significant global public health crisis, intricately linked to multiple metabolic disorders, including type 2 diabetes and cardiovascular diseases. Researchers from the Department of Pharmacology and Pharmacy at the University of Hong Kong (HKUMed) have made a groundbreaking discovery, identifying the mitochondrial protein Chchd10 as a critical regulator of adipose tissue homeostasis, presenting fresh avenues for tackling obesity and its associated health complications. Their illuminating findings have been published in the prestigious journal Advanced Science.
Importance of Adipose Tissue
The wellbeing of adipose tissue is fundamental for sustaining the body's energy balance and overall metabolic health. When individuals consume excess calories, adipose tissue undergoes remodeling, leading to an increase in both the number and size of fat cells. However, if this remodeling is not properly balanced, it can precipitate obesity and a host of metabolic disorders.
Research demonstrates that the location of fat accumulation in the body can significantly influence metabolic health. For instance, excessive visceral fat is known to pose higher risks for metabolic complications, while subcutaneous fat is generally considered less detrimental. Consequently, understanding the intricate molecular mechanisms behind adipose tissue homeostasis is pivotal for developing effective obesity interventions.
Groundbreaking Research on Chchd10
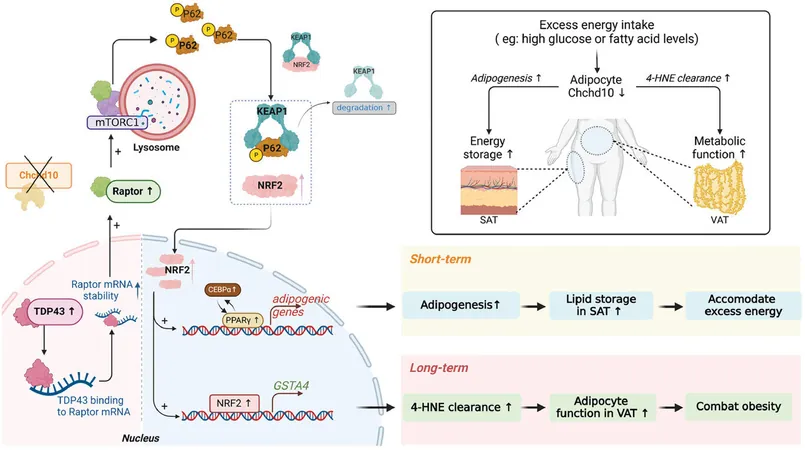
The HKUMed research team conducted extensive investigations into Chchd10's role in adipose tissue remodeling, utilizing a methodology involving high-fat diet (HFD)-induced wild-type mice and adipose tissue-specific Chchd10 (AT-Chchd10)-deficient mice. They discovered that in HFD-induced mice, Chchd10 levels in white adipose tissue—responsible for energy storage and metabolic regulation—were significantly diminished. This drop in Chchd10 levels expedited the formation of subcutaneous fat, enabling the mice to more effectively conserve energy during periods of high-fat consumption.
Conversely, the absence of Chchd10 led to elevated levels of GSTA4, a protein found in visceral fat that plays a crucial role in clearing harmful lipid peroxidation products and preventing cellular dysfunction. The study highlighted that reduced Chchd10 levels activated the NRF2 signaling pathway, fostering subcutaneous fat formation and enhancing GSTA4 expression.
Implications of the Findings
The implications of this research are profound. It identifies Chchd10 as a novel regulatory factor essential for maintaining adipose tissue stability and balance. The downregulation of Chchd10 not only promotes subcutaneous fat formation but also regulates antioxidant capacity primarily in visceral adipose tissue, thus mitigating the risk of obesity and metabolic disorders linked to dietary habits.
Moreover, evidence suggests that knocking out Chchd10 in adipose tissue during weight gain stages related to a high-fat diet led to a significant reduction in visceral fat mass. This finding opens the door to the possibility that manipulating Chchd10 levels could serve as an innovative therapeutic strategy for obesity management.
Professor Ruby Hoo Lai-chong, an Associate Professor at HKUMed, emphasized the significance of this study, stating, "This research sheds light on the molecular pathways that play a vital role in regulating adipose tissue homeostasis and their implications in obesity management. Targeting Chchd10 and its associated pathways may pave the way for developing new therapeutic approaches to combat diet-induced obesity and enhance metabolic health."
In addition to this groundbreaking work on Chchd10, Professor Hoo's team has previously explored other critical roles in metabolic conditions, such as their findings on tissue-resident memory T (TRM) cells and their involvement in type 1 diabetes. Their research underscores a wider connection between immune response and metabolic diseases, highlighting the promise of novel interventions targeting metabolic dysfunctions tailored to specific adipose tissue types.
Conclusion
As the battle against obesity intensifies globally, insights from this research could revolutionize approaches to managing not only obesity but also the complex associated metabolic disorders.


 Brasil (PT)
Brasil (PT)
 Canada (EN)
Canada (EN)
 Chile (ES)
Chile (ES)
 Česko (CS)
Česko (CS)
 대한민국 (KO)
대한민국 (KO)
 España (ES)
España (ES)
 France (FR)
France (FR)
 Hong Kong (EN)
Hong Kong (EN)
 Italia (IT)
Italia (IT)
 日本 (JA)
日本 (JA)
 Magyarország (HU)
Magyarország (HU)
 Norge (NO)
Norge (NO)
 Polska (PL)
Polska (PL)
 Schweiz (DE)
Schweiz (DE)
 Singapore (EN)
Singapore (EN)
 Sverige (SV)
Sverige (SV)
 Suomi (FI)
Suomi (FI)
 Türkiye (TR)
Türkiye (TR)
 الإمارات العربية المتحدة (AR)
الإمارات العربية المتحدة (AR)